お知らせ・お役立ち便覧 NEWS
有機溶剤
2025.05.09
アセトンとは?特徴を初心者にもわかりやすく解説

マニュキュアを落とす除光液の主成分として知られるアセトンについて、化学の知識がない方でも理解していただけるように、わかりやすく丁寧に解説します。
目次
アセトンとは?
アセトンとは、ケトン系の有機溶剤の一つで、ジメチルケトンや2-プロパノンとも呼びます。
ケトン系の有機溶剤は、ケトン基という官能基《R−C(=O)−R’ (R, R’ はアルキル基など)》を持っているものの総称で、アセトンはケトン系有機溶剤の中で最も単純な構造を持っています。
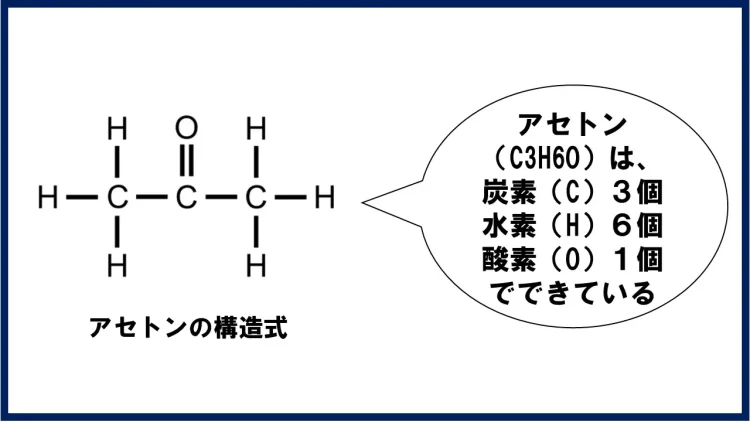
化学式はC3H6Oで、炭素(C)が3個、水素(H)が6個、酸素(O)が1個から成ります。
アセトンの特徴は以下の通りです。
| 項目 | 概要 |
| 名称 | アセトン |
| 別称 | ジメチルケトン、2-プロパノン |
| CAS番号 | 67-64-1 |
| 化学式 | C3H6O |
| 引火点(℃) | -17 |
| 発火点(℃) | 540 |
| 沸点(℃) | 56 |
| 比重 | 0.79 |
| 有機溶剤中毒予防規則 | 第2種有機溶剤等 |
| PRTR法 | 非該当 |
| 消防法 | 第1石油類水溶性 |
アセトンは水にも油脂にも溶ける(親水性と親油性がある)ため、さまざまな用途に用いられます。しかし、上記のように引火点(火を近づけたときに燃え始める温度)がマイナス17℃と大変低く、火災の恐れがあるため消防法により規制している他、毒性があるため有機溶剤中毒予防規則(有機則)でも規制しています。
アセトンの法規制
 アセトンは有機溶剤中毒予防規則や消防法などで規制しています。
アセトンは有機溶剤中毒予防規則や消防法などで規制しています。
消防法による規制
アセトンは常温でも火元があれば簡単に燃えてしまうほど火が着きやすく、消防法では第1石油類水溶性液体に分類しています。
消防法の規制を受けずに保管できる量である指定数量は、400Lです。
また、危険物倉庫がなくても保管できる少量危険物(指定数量の1/5)は80Lです。
詳しい規制の内容は、別記事「消防法とは?該当する有機溶剤、指定数量の考え方などもわかりやすく解説」で詳しく説明しておりますので、併せてご覧ください。
アセトンの毒性と有機溶剤中毒予防規則(有機則)による規制
有機則とは、有機溶剤を取り扱う上で作業者の安全を守るための法令です。
有機則では、指定した有機溶剤44種を毒性が強いものから順に第1種、第2種、第3種に区分しており、アセトンは第2種有機溶剤に該当します。
アセトンの具体的な毒性としては、眼刺激、生殖能または胎児への悪影響の恐れの疑い、眠気やめまいの恐れ、呼吸器への刺激の恐れ、長期または反復曝露で血液の障害の恐れ、飲み込みや気道に侵入すると有害の恐れがあるとされています。
アセトンを取り扱う際は、有機則により、専用の保護具を使用することや、換気設備を設けること、資格を持った有機溶剤作業主任者を選任することなどさまざまな規制が掛かります。
詳しくは別記事「有機溶剤中毒予防規則(有機則)とは?わかりやすく解説」で丁寧に説明しておりますので併せてお読みください。
アセトン不要のジェルネイルリムーバー
アルミホイルやアセトンを使わない、新技術のジェルネイルリムーバーはこちらです。
アルミ巻きが不要で、セルフオフも可能。アセトンによる肌の乾燥も防ぐことができます。
身近なアセトン
 アセトンは、水、油、ほとんどの有機溶剤と混ざるため、本来混じり合わない液体を混ぜ合わせるためのカップリング剤になります。また乾きやすいため、実験器具の洗浄などにも使用します。
アセトンは、水、油、ほとんどの有機溶剤と混ざるため、本来混じり合わない液体を混ぜ合わせるためのカップリング剤になります。また乾きやすいため、実験器具の洗浄などにも使用します。
このほか、さまざまな用途で利用されており、私たちの身近にもアセトンを使用したものがあります。
たとえばマニキュアやネイルポリッシュなどのネイルカラーを落とす除光液(リムーバー)の主成分はアセトンです。
また、接着剤や塗料の溶剤、瞬間接着剤の剥がし液などにも含まれます。
アセトンの販売について(2023/03/15追記)
アセトンは引火性が高く、また火薬の原料となるため、一般には販売していません。
三協化学のアセトン関連製品
弊社のアセトン関連製品には以下のようなものがあります。
ファインソルブシリーズ⋯塗料やインクの除去、樹脂の溶解
メタルクリーナーシリーズ⋯切削油や潤滑油の洗浄
アセトンに関する疑問質問は下記リンクまでお気軽にお問い合わせください。
アセトンに関する質問
※2024年11月19日更新
以前、ページのコメントに寄せられた疑問・質問とその回答をご紹介します。
(※一部編集・抜粋しております)
質問No.01~10
※「▼」をクリックすると質問が表示されます。
住宅施工業者が、改修工事後に養生テープを剥がしたら、床のワックスの一部が一緒に剥がれて‟部分剥離”してしまい、ダメージ部分と非ダメージ部分との間に‟色具合の差”や‟僅かな段差”などの不具合が生じてしまいました。
業者は「アセトン洗浄して床面全体をきれいにした後で、水性ワックスを塗って原状回復したい」と言っていますが、この方法は有効でしょうか?
(業者は一応、部分的なパッチテストみたいなことをして「結果は良好だった」と言っています)。引火性や有毒性も高いようなので、ちょっと心配しています。
一般住宅の床面全面をアセトン洗浄するときの‟注意事項”などがありましたら、ご教示ください。(作業中の注意もさることながら、将来の‟ワックス塗り直しメンテ”時の注意事項にも興味を持っています。)
また物件は‟賃貸用物件”で、現在は新規入居者募集に向けて原状回復メンテ中なのですが、問題の‟原因物質”は「かなり長期にわたって室内に残留する」と考えてよろしいでしょうか?
(有害物質が室内から消散するのに、大体どのくらいの時間がかかるものでしょうか?)
ワックスの種類がわからないので、テスト結果から見て判断せざるをえない状況です。パッチテストで良好だったのであれば有効ではあると思います。
ただ、おっしゃる通りアセトンは引火性や毒性があるため好ましくありません。アセトンはシックハウス症候群の原因物質の一つでもあるため、ワックス塗り直しメンテナンスの後の物件で症状が現れるケースも起こりえます。(アセトンがシックハウス症候群で長期的に残留する場合は、壁紙などの接着剤として使用された場合です)
アセトン以外のものでも出来ないか試されることをお勧めいたします。一般的にワックスは炭化水素ですので、揮発の遅い炭化水素類で試されるのが良いでしょう。
販促のようになってしまい恐縮ですが、弊社三協化学の製品ですと、メタルクリーナー2000やメタルクリーナー1700で一度お試しになってみてはいかがでしょうか。また液体のまま使用した場合、長期にわたって残留せず、1日ほどしっかり換気すれば残留物は、ほとんどなくなります。
ただし毒性自体は液体として使用しても接着剤として使用しても同じですので、今後は見直しを検討するのも良いかと思います。
アセトンで消毒液は作れますか?
アセトンのウイルスや菌に効いた、というデータは弊社では持っていないことと、アセトンの危険性の高さから、弊社ではお勧めはできません。
もし効果があったとしても、使用者に事故や害が及んでしまえば、元も子もありません。
エンボスシートに、油性のマジック(赤色)で落書きされました。既に3か月経過しています。
アセトンで除去出来ますか。アセトンと洗濯ノリで湿布、ラップで湿布、これで除去出来ますでしょうか。
また湿布時間等は、どれ位かかりますか。その前の除去出来ますでしょうか。
近日中に作業をしたいのですが、他に除去出来ますでしょうか。
通常の油性マジックでございましたら、アセトンで拭き取れば落ちるかと思います。
妊婦がアセトンを使用すると胎児に影響があるのでしょうか?母体にも良くないのでしょうか?
液体のアセトンを使ってアルミや銅板の脱脂(拭き取り)をする仕事をしています。現在私は妊娠3ヶ月です。市販のマスクを着用しております。広い空間で1日1~2時間程度使用しています。
また、IPA溶剤も同じように使用しています。これもまた胎児、母体ともに影響はありますか?
アセトンは女性則には該当しませんが、作業中の空気に含まれる溶剤の濃度によっては装着されるマスクに問題があるかもしれません。
市販のマスクとのことですがそれは防毒マスクでしょうか。
体にあまりよくないものですから、防毒マスクをお勧めいたします。
また、IPAもアセトンと同様、市販の花粉症対策などに使う一般的なマスクではなく、防毒マスクを着用しての使用をお願いいたします。
車のヘットライトカバーがプラスチックでできていますが、黄色く変色したのでアセトンで取れますか教えてください。
プラスチック自体が変色してしまっているので、アセトンで拭いても黄ばみを取ることはできません。
弊社の製品ではありませんが、他社製品で黄ばみ取りという製品がホームセンターや車用品を取り扱っているお店で販売されていると思いますので、そちらをご使用になられることをお勧めいたします。
洋服に瞬間接着剤を付けてしまいました。
アセトンの原液で取れるとインターネットに出ていますが、本当でしょうか?
固いものに付着した場合は落ちるのですが、布のような繊維の隙間に入った場合は、完全には落ちない場合があります。
また、布によっては、繊維や色素へのダメージも発生する恐れがあります。
黒板に油性のサインペンで線を書いています。
これをアセトンを主成分とする百均の除光液で拭き取ろうと考えておりますが、黒板の表面にダメージ等はないのでしょうか?
少し不安になり、質問をさせて頂いております。
除光液の成分は黒板表面の塗料までも落としてしまう可能性が非常に高いです。
上から黒板塗料で塗り直した方が良いかと思います。
革靴に塗料をこぼしてしまったのですがアセトンで塗料を落としても革靴が痛んだり、色落ちなどしないですか?
塗料が溶けるような溶剤ですと、革靴の色を落としてしまう可能性が非常に高いです。
革靴の色付けも塗料によって施されていることが多いので、どちらかを残してどちらかだけ落とす、というのはデリケートな作業になってしまいます。
ジェルネイルをとるためにアセトンを染み込ませたコットンをホイルでおおった状態のまま長く放置していたため、指先が脱脂しています。
お風呂では指先がすぐフヤケたり、指先が常に脱脂したような感覚です。元に戻らないのでしょうか?
皮膚は一定期間で代謝されますので元の状態に徐々に戻るかと思います。
それでも気になる場合は、皮膚科の受診をお勧めします。
コンクリートの床の上に購入したタイヤを半年以上置いておいたら、黒く丸い染みが着いてしまいました。アセトンで落ちますでしょうか?
アセトンで大まかには落とすことができますが、コンクリート表面の細かい穴に入り込んでしまった汚れは落ち切らない恐れがあります。
また、アセトンは引火性が高い溶剤ですので、あまりお勧めはできません。
質問No.11~20
※「▼」をクリックすると質問が表示されます。
エアコンの配管洗浄に使用することは出来ますでしょうか?方法は、ある容器に入れて、窒素ガスで配管に送り込み、油分等を取るという方法です。
ガラスや、銅管、アルミ等に腐食や変色などは出ないでしょうか?
物理的には可能ですが、アセトンは有機溶剤中毒予防規則が該当致しますので、アセトンの代替品の使用をお勧めします。
車のシートに瞬間接着剤と工業用のボンドを両方を付けて仕舞いました。アセトン原液とシンナーでで拭いたのですが取れません。
他に方法は有りますでしょうか。
自動車のシートは布製でしょうか?布製の場合は生地の隙間に流れ込んで硬化しているため、生地自体を溶剤に浸す必要があります。
また素材によっては溶剤に弱いものもありますので、目立たないところで一度試してから使用してください。
革製の場合、溶剤で拭けば落ちると思いますが革の色も一緒に落ちてしまう可能性があります。こちらも目立たないところで一度お試しください。
トルエン30%配合のダイアボンドとシンナーを使う作業をしています。
ボンドを注射器に入れて、部品に塗布するのですが、ハンダ作業用の吸気ダクトは有りますが、マスクは有りません。
1日7時間作業で一月くらい作業してますが、後々体調に影響が出てくるのでしょうか?
局所排気装置がついていれば、マスクの着用は義務ではありません。
体に影響があるかは、使用環境に加え個人差もございますので、体調に不安があるようでしたらお早めに医療機関を受診してください。
ステンレス製品にニトリルゴムが装着されていてニトリルゴムのみを融解させ取り除く為にアセトンを用い様と考え試験した結果ゴムが劣化し除去は出来そうなのですが、作業の際に入れ物として樹脂トレーでは変型等が発生してしまうでしようか。
変型しない樹脂が有りましたらご教授願います。又、ステンレス自体(SUS303系)に悪影響は有りませんでしょうか。
容器については樹脂トレイよりも金属製の容器を推奨いたします。(樹脂トレイは、アセトンが溶かしてしまう恐れがあるものが含まれている可能性があるため)
ステンレスに関しては問題ありません。
アセトンに浮く材質はありますか?廃棄処理時に困っております。
廃棄用容器にアセトンを含んだ廃棄溶剤を溜めて処理をするのですが、容器の中身がどれくらい入ったか廃棄溶剤を入れる穴からは見えないので時々溢れてしまいます。
そこで、穴の上からでもどのくらい溶剤が溜まったか簡単に目視できるように、アセトンに浮く材質のウキ的な物を使いたいと考えています。以前、釣り用のウキを購入して試してみましたが沈んでしまい失敗しました。(アセトンの比重は水より軽い為。)
また、液面センサは高価であり手が出しにくいためアセトンに浮く材質を教えて頂ければ大変助かります!
アセトンより比重の軽い物質はありますが、侵してしまうおそれがありますので、ポリエチレンの小さい容器などの中に空気を入れて「浮き」になさることをお勧め致します。
フロアコーティングの補修時に業者がアセトンで床の汚れを拭き取っていました。その際にエアコンを稼働しました。エアコンに何か影響はあるのでしょうか?
エアコンの空気の対流がアセトン臭をまとって、部屋中が臭くなる…ということはありますが、稼働させていればにおいも消えていきますので、ご安心ください。
娘が毎日八時間程度、アセトン使用しガラスなどの拭き取り作業をています。
空調設備は整っているようですが、市販されている不織布のマスクを家から着けてきそのまま帰宅します。
作業中は、防毒タイプや違ったマスクの方がいいのでしょうか?
空調設備がどのようなものかにもよりますが、(局所排気やプッシュプル排気設備であれば、法的には問題ありません)
もし換気扇があるだけ、であるなどの場合、防毒マスクなどの保護マスクの着用が法律で義務付けられています。
一度空調設備について、ご確認ください。
>>換気装置に関する詳しい情報ページ
長年しまってあった抹茶茶碗に、茶渋がしっかり付着していました。
それならアセトンを染ませた綿棒で、クルクル拭きとると良いとの助言をもらいましたが、このような使い方で茶渋が取れるのでしょうか。
また茶碗への影響はどうでしょう。
また、抹茶茶碗は繊細なものが多いように思い、漂白剤など躊躇しておりましたが試してもよろしいでしょうか。
アセトンでも落とすことはできますが、アセトン自体の入手がしづらい状況にありますので、酸素系の漂白剤で十分に落としうるかと思います。
また、釉薬がしっかりかかっているお茶碗でございましたらメラミンスポンジなども有効です。抹茶茶碗に金属系の装飾がついていなければ酸素系漂白剤で問題ありません。
もし金属系の装飾がついている場合はアセトンの方がよいです。
アセトンを小分けする容器について知りたいのですが、30mlくらいの容器を探しています。
素材など何でしたら溶けない、揮発しないなど教えて頂けないでしょうか?
アセトンの容器でベストなのは金属です。ブチルゴムなど、アセトンに侵されない素材のパッキンがついた金属容器をお勧め致します。
樹脂容器では、ポリエチレンやポリプロピレンなどでしたら大丈夫です。
容器が半開きであったり、隙間があったり、穴が開いていればどんな容器でも揮発したりこぼれたりしてしまいますので注意は必要です。
マジックインキが乾燥してしまいました。アセトンを入れて、元に戻す事が出来ますか?
アセトンではタイプが違うため元に戻りません、また危険性を伴いますのでおやめください。
なんの溶剤が合うかはインクメーカーごとに異なりますので、メーカー様にご確認ください。
質問No.21~30
※「▼」をクリックすると質問が表示されます。
仕事で建築用養生シートや防音シートなどをクリーニングしているのですが、ペンキ汚れやタールなどを落とすためにラッカーシンナーを使用しています。
しかしシンナーのあの臭いが不快なので、他にいい汚れ落とし剤がないかと探しております。
値段や、汚れの落とし具合、安全性など、ラッカーシンナーと比較して他に考えられる良い溶剤はありませんでしょうか?
溶剤も、溶解力が強い溶剤は総じてにおいが強い傾向にあります。
安全性に関しては、どこを重視するかによって、ご案内できる製品が異なってしまいます。
(例えば引火性に関してなのか、あるいは溶剤の毒性に関してなのか、など)また、どうしてもラッカーシンナーの方が安価であるということもあり、どこを重視してどこを妥協するかにもよってご提案内容が変わってしまいますので、
もし弊社三協化学の製品でそういったものを探してみたい、試してみたいということであれば
https://www.sankyo-chem.com/contact/
上記のお問い合わせフォームよりお問い合わせ頂けますと幸いです。
窓ガラスに貼ってるフィルムの金属系のコーティング材を剥がしたのですがアセトンで除去出来ますか?
今貼られているコーティング材の成分が分かりませんと、正確なお答えは出せないのですが、金属はアセトンで溶かすことができないので、難しいかと思われます。
以下の場合、溶剤で考えた時は何の溶剤が一番適しているでしょうか。
新築の工事現場で、コンクリート素地仕上げの躯体階段を数ヶ月養生していました。
作業者は跡が残らないようにと青のマスキングテープを貼り両面テープ、プラベニヤの順で養生していたようですがマスキングの跡が黒く残ってしまい、ペーパー掛けやシンナーでも落としきれていません。
次に試す予定のものはアセトン、ジッポーオイルなどです。
おそらくジッポーオイルで落とすことができるかと思いますが、ジッポーオイルでお試しになって、ダメだったらアセトンで落とされるのが良いかと思います。
どちらも引火性が高いので、火気にはくれぐれもお気をつけになって、しっかり換気をしながらご使用ください。
アイロンで熱を加えた布用ボンドは、アセトンの原液で取ることは可能ですか?
リサイクルショップで購入した野球のユニフォームのズボンにアイロン圧着の衝撃パットが変な位置に着いていたのでアイロンで熱を加えて剥がしたらどうやらアイロン圧着ではなく布用ボンドで貼りつけてありました。
パットは簡単に取れたもののユニフォームにはボンドの跡がついたまま。
100均の除光液である程度は取れましたがまだ繊維の中にはボンドの跡があります。
除光液は、アセトンが主成分のものや、酢酸ブチルが主成分のもの、エタノールが主成分のものなどありますので、お手持ちの除光液で落ちたのであれば、そちらの除光液の成分をご確認頂き、そちらの原液をお使いになるのが良いかと思います。
どちらの有機溶剤も、引火点が低いため、使用時は火気などにお気をつけて、また、しっかり換気をなさってゴム手袋着用のもとご使用ください。
合皮生地の表面に付着した寸法合わせのマーキングを除去するのに、ウェスにアセトンを軽く染み込ませて拭き取っております。
光沢が出ないようにしたいのですが、何か方法などありましたら教えていただきたいです。
マーキングは除去できるのですが、アセトンで拭いた箇所は合皮生地の表面にやや光沢ができてしまい困っています。
表面が溶けることで、滑らかになり光沢が発生しているものと思われます。アセトンよりも溶解力の低いもので拭かれることをお勧め致します。
マーキングがどのようなものかわからないので、漠然とした回答で申し訳ありません。
物置を整理していたら古い棚から試薬アセトンが数本出てきました。以前父が何かで使用してしたものと思います。どうのように処分したらよいのでしょうか?
危険物は、管轄の自治体様によって、扱いが異なりますので、お住まいの管轄の自治体様へお問い合わせいただくのが確実かつ最短でございます。
スニーカーのソールのペイント(サイドソール)はアセトンで落とすことはできますか?
塗料によっては落とすことができますが、同時に素地を痛めるおそれがございます。
もしお使いになる場合は、目立たないところで少しだけお試しになってから、ご使用ください。
また、ご使用時は火気に十分ご注意の上、しっかり換気、保護具をお使いの上ご使用ください。
庭のコンクリートに人工芝が貼り付けてあります。水はけが悪く青のりが発生しているので剥がしたいのですが、強力な接着剤で剥がすのに苦慮しています。
スクレイパーを試しましたが力が要るので、もっと楽に剥がしたいです。
アセトンは有効ですか?有効の場合、6m×6mほどの広さでどのくらいの量が必要ですか?
接着剤がどのような成分のものか分からないので、剥離剤の選定ができません、申し訳ありません。
また、仮にアセトンで溶けたとしても、今の時期でしたらアセトンは揮発性が高く、すぐに乾いてしまうため、作業がしづらいかなと思いますので、あまりお勧めできません。
金属の表面を清掃する場合、アセトンとIPAの違いは何でしょうか?
アセトンは、IPAに比べ、脱脂能力や樹脂溶解力の優れた溶剤です。
アセトンは今入手しづらくなっており、また、引火点がIPAよりも高いので、IPAが選ばれるケースもあります。
また、金属表面にアセトンでは溶けてしまう樹脂などが付着しており、それは落としたくない場合、IPAが使われます。
スチールベアリングの脱脂には普段からアセトンを使用しているのですが、セラミックベアリングの脱脂にアセトンを使用してもセラミックボールに影響は無いでしょうか?
セラミックボールでございましたら、アセトンは影響ございませんのでご安心ください。
質問No.31~40
※「▼」をクリックすると質問が表示されます。
赤ちゃんへの影響について、質問です。
高さ90センチのテーブルの落書きを消そうとアセトン除光液を使ってしまいました。取れず、テーブルは、はげ、どうにかしようと何度も使ってしまいました。
部屋には7ヶ月の赤ちゃんがおりハイハイしたりつかまり立ちしたりしていました。時々テーブル近くに来たり、泣いて、抱っこして駅をふき取ったりもありました。
換気の必要性に気づいたのが3、40分後です。1時間くらい、もしかしたらこい濃度の揮発したアセトンをすってしまったかもしれません。
脳や、その後の発達に影響ありますか
耐性には個人差がございますので、かかりつけのお医者様にご相談ください。
自家用車のプラスチック部品の補修に瞬間接着剤を使用した時にボディに接着剤がたれてしまいました、取り除く為にアセトンは有効でしょうか?
アセトンを使用する際は、自動車の塗装を落としてしまうことがありますので、接着剤が付着してしまった部分以外に付かないように十分注意ください。
バイクの燃料タンクの吸い込み口の網目部分にガソリンのガム質がこびりついて、ガソリンが流れません。
目視出来ないので推測ですが、アセトンをタンク内に入れて漬けておく事でガム質は溶けるでしょうか。
一度分解なさることをお勧め致します。
①バイクの燃料タンクのつまりの原因は、ガム質だけでなく錆なども原因となるため、つまりの原因を明らかにするため
②汚れを一緒に流さないほうが良いので、分解した状態での洗浄をお勧めするため
アセトンは引火性が高いのでキャブレタークリーナーをご使用になることをお勧め致します。
合成樹脂エマルジョンの接着剤に、アセトンが有効な剥離用剤となるでしょうか?
オフィスに特化した産廃業者です。最近スチールパーティション(表面塗装鋼板の裏に接着剤を塗布して石膏ボードが貼り付けている構造)の引取りが多く、鋼板と石膏の剥離に苦労しております。メーカーに問合せところ、合成樹脂エマルジョンのペガールLW6255という接着剤を使用しているようですが、アセトンが有効な剥離用剤となるでしょうか?
また、鋼板と石膏がしっかり密着しており、アセトンを浸透させる良い方法があればご教授くださいませ。
接着剤にアセトンを浸透させればよいのですが、接着剤の露出している表面積が少ないため、正直難しいです。
大量の発泡スチロールがあって、アセトンに漬け込んだらドロドロに溶ける動画を見て、この方法で処理したいと考えているのですが、溶けた発泡スチロールや残ったアセトンの処理はどのようにしたらよいでしょうか?
処理については管轄の自治体様によって、推奨方法が異なるため、ご準備の前にご確認されることをお勧め致します。
また、アセトンは引火性液体ですが、発泡スチロールは電気を帯びやすく、溶かそうとした際に生じる静電気によって引火する危険性が高いため、注意が必要です。
賃貸マンションのバスタブ(多分FRP)の黒ずみか落ちなくて困ってます。
恐らく20年以上は使用されていて、クレンザー・重曹・市販のバスタブクリーナー等では歯が立ちません。
アセトンをキッチンペーパーに染み込ませ、ラップすれば効果は期待出来るでしょうか?
知見がないため、はっきりとしたことは申し上げられませんが、おそらくアセトンでは効果はないかと思います。
アセトンとMEKに大きな差(洗浄力やオイル溶解度など)はあるでしょうか??
内燃機の整備にアセトンを使用しています。主な用途は脱脂としてエンジンオイルで汚れた部分の洗浄、ほかに燃焼室内に付着したカーボンの除去に使用しています。(ウエスに染み込ませての拭き取り)
多量に使うわけではないのですがそこそこ高価なアセトンに対して同業者が安価なメチルエチルケトンを使用している話を聞きました。
アセトンもMEKも価格は大きく差はありません。
ただMEKは毒物及び劇物取締法に該当しますので、購入の際は販売店に使用目的の説明と印鑑が必要になります。
洗浄力も大きな差はありませんが、アセトンの方が乾燥性が早いので、ご使用方法によってはMEKの方が洗浄しやすいかもしれません。
アセトンの長期保存に関しまして、1番影響を受けない、1番長期の耐久性がある以下のうち、素材はどれになりますか。
鉄
ブリキ
スチール
ステンレス
塗装などで使われる密封型の丸管で予定しております。
ステンレスが最も影響を受けません。
挙げていただいたものは全てアセトンでは侵されませんが、ブリキや鉄(スチール)はアセトン関係なく空気中で錆びるため、長期間置いておくことは望ましくありません。
作業中1リットル位トルエンが体や服に付着しました。作業優先なので続行しましたが体調不良気味です。
会社に知られないよう、病院にいかずに症状が良くなるのを待つつもりですが、耐性が低い場合時間がかかりますか?
有機溶剤だが、特別に対策せずとも大丈夫との話しを聞いたのですが。
人によって耐性には個人差がありますので、他の人が同じ状況で大丈夫だったからといって、自分も大丈夫であるとは限りません。
ご不安であれば、かかりつけのお医者様にご相談下さい。
また、トルエンは有機則にも該当する有機溶剤なので、必ず防毒マスクやゴーグル、手袋を着用し使用してください。
アセトンを含有したアルコールスプレーを肌につけて良いかでしょうか?
個人で香水を作る為にマルトールの結晶を購入したのですが、液状する際にエタノールでは溶けなかったので、アセトンで溶かすことを検討しています。
化粧品、ネイル用品として売られているアセトン100%の商品でマルトールを50%に希釈し、最終的に8%程マルトールを含有したエタノールスプレー(香水)を作ろうと考えています。この場合、肌につけても大丈夫でしょうか?
化粧品は弊社の分野ではありませんが、アセトンをつけたままにするのはお勧めできません。
質問No.41~
※「▼」をクリックすると質問が表示されます。
アセトンをガラス瓶に入れて長期保存はできますでしょうか。
アセトンは揮発性が高いので、密閉できる蓋を使用しての保管であれば問題ありません。
かつ、蓋やパッキンのゴムなどが、しっかりアセトンに耐性のある素材かどうかが重要となります。
アセトンに耐性がないと、保管中に密閉できなくなり、揮発してしまうおそれがございますので、しっかり材質を確認の上、保管ください。
ジェルネイルをオフするのに密閉容器にアセトンを加温させ蒸気をあててオフしています。
その際密閉容器から漏れ出てしまった蒸気となったアセトンは、何か人体に影響はでますか?
「密閉容器」というのは密閉されているはずなので、中の気体や液体が漏れ出すことはありません。
逆に、中の気体や液体が漏れ出すようであれば、それは「密閉容器」ではないため、保管している間に揮発して、近くに火気などがあれば引火の可能性があったり、誤って吸い込んでしまったりして危険です。また、アセトンの加温は、常温時よりも更に引火性が高くなりますし、保護具の着用がない場合、蒸気を吸い込んでしまい、危険です。
具体的な影響に関しては個人差があるため、一概に申し上げることはできませんが、メーカーが作成しているSDS(安全データシート)をご参照の上、正しくお使いいただけますようお願い申し上げます。弊社のアセトンのSDSは下記URLよりご参照いただけます。
https://www.sankyo-chem.com/wp/wp-content/uploads/acetone.pdf
以下の状況で、1.5本分くらい除光液を使用しました。健康被害が心配です。
2日前友人が私の家の駐車場でバイクのオイルを交換する際にオイルを大量にこぼしてしまいました。
それを水で流してしまい、その処理の仕方が間違えだと気づき二人でできる限り排水溝に流れてしまったオイルをキッチンペーパーで拭き取りました。
その後に地面がキラキラしているのに気づき放って置いたらシミになって残ると友人が言っていたのでキラキラを処理するために除光液を使いました。1.5本分くらい使っていたと思います。
家に帰ってアセトンが危険なものだと気づきました。
マンションなので他の方の健康被害が心配です。今のところみなさん異常はないようなのですが大丈夫でしょうか?
アセトンは揮発性が高い物質ですので、屋外でしっかり換気がされる環境であれば、じきに揮発しアセトンを含んだ空気も入れ替わります。
溶剤の人体への影響は、個人差もありますので、ごく微量でも気分が悪くなってしまう方、多少吸い込んでしまっても大丈夫な方いらっしゃいますので、ご不安な場合はかかりつけのお医者様へご相談なさる事をお勧め致します。
アセトンの効果についてお伺いしたいのです。
ポリエステル100%の白Tシャツに昇華インクでプリントしたのですが熱転写したプリント用紙の角に汚れがあったようで無地の部分に汚れもプリントされてしまったのですが、液体のアセトンを刷毛で塗布したら綺麗に取れますか?
注意事項などあればご教授頂きたいです。
プリント用紙の角にあった汚れが、昇華インクかどうか分かりませんが、昇華インクには染料が使われているため、アセトンでは難しいかもしれません。
もしご使用になる場合、アセトンは揮発性が高く、また引火性も高いので、火気や静電気にはくれぐれもご注意いただき、目や口に入らぬよう保護していただく、また気化したアセトンを吸い込まないようしっかり換気してご使用ください。
アセトンをスポンジ等に染み込ませて塗料を落とすことは可能ですか?
また、他の用具で使用可能なものがあれば教えて欲しいです。
アセトンに溶ける塗料ならば塗料を落とすことができます。
アセトンに溶解せず染み込ませて拭き上げることができる用具なら使用できます、たとえば綿でできたウエス等が使用できます。
カテゴリーから探す
キーワードから探す
